Members and suppliers: Join our online community
Access our full library of resources, register for events and webinars and share audits and other assessments
Access our full library of resources, register for events and webinars and share audits and other assessments
Our resource library is made available freely to all in order to advance responsible supply chain practices. Here you will find information and guidance on responsible supply chain issues and the way that the PSCI works. If you have any suggestions for improvements or additions to our resources please contact us.
Pharmaceutical and healthcare companies run thousands of audits of their suppliers’ sites annually. Audits typically require between two to four days of focused, on-site effort from both the auditor and the supplier. PSCI members work together to share these audits, in support of greater transparency and valuable efficiencies that benefit both members and suppliers.
This report shares our analysis of findings from PSCI audits available on our platform. Each year, we review these in line with the PSCI Principles to identify trends, highlight improvements, and share recommendations. PSCI work with member companies, suppliers, and regional partners to turn analysis into practical guidance, training, and peer learning that suppliers can implement quickly. Overall, this analysis provides deep insight into supplier practices within the pharmaceutical and healthcare industry and suggests where collaborative efforts will have the greatest impact, guiding us on developing targeted resources and trainings.
This year’s report analyzes 2,046 findings from 228 supplier audits uploaded to the PSCI platform in 2024. These findings are categorised into 428 Governance & Management Systems findings, 26 Ethics findings, 273 Human Rights findings, 1,098 Health & Safety findings, and 221 Environment findings, offering a comprehensive overview of supplier practices and opportunities for improvement. The report includes a summary of findings across key topics, as well as a regional analysis looking at patterns across China, India, US, Western Europe, and other regions represented in this year’s uploads, presenting recommended actions for suppliers.
The commercial and sales teams in the pharmaceutical industry spend a significant amount of time traveling to meet healthcare providers, distributors, and customers across diverse geographies. Road safety is the number one risk in India and road incidents directly impact personal safety, business continuity, and customer engagement.
This session will cover key aspects of road safety including safe driving practices and behaviours, managing fatigue and stress on the road, and the importance of avoiding distractions while driving. Demonstrating leadership in road safety strengthens employee engagement, corporate reputation, supports ESG commitments, and contributes to India’s National Road Safety Mission. By participating, you can help your organizations drive collective action towards a safer, more responsible, and resilient pharmaceutical ecosystem.
Speakers:
Mahesh Chandak
Head of SSHE - South Asia and Global Road Safety Lead, Bayer
Rajani Chavan
SSHE Enablement Lead, South Asia and India Road Safety lead, Bayer
The associated slides can be downloaded here
The commercial and sales teams in the pharmaceutical industry spend a significant amount of time traveling to meet healthcare providers, distributors, and customers across diverse geographies. Road safety is the number one risk in India and road incidents directly impact personal safety, business continuity, and customer engagement.
This session will cover key aspects of road safety including safe driving practices and behaviours, managing fatigue and stress on the road, and the importance of avoiding distractions while driving. Demonstrating leadership in road safety strengthens employee engagement, corporate reputation, supports ESG commitments, and contributes to India’s National Road Safety Mission. By participating, you can help your organizations drive collective action towards a safer, more responsible, and resilient pharmaceutical ecosystem.
Speakers:
Mahesh Chandak
Head of SSHE - South Asia and Global Road Safety Lead, Bayer
Rajani Chavan
SSHE Enablement Lead, South Asia and India Road Safety lead, Bayer
The recording for this webinar can be accessed here
The PSCI was delighted to exhibit at CPHI Frankfurt 2025 as an Official Sustainability Partner of the conference. We engaged with Members, Supplier Partners, and other Partners all working towards building responsible supply chains.
Check out our highlights video and see you next year at CPHI Milan!
The 2023 revision of the PSCI Principles (PSCI Principles v3.0) was approved by the membership on Tuesday 23rd May 2023, at the Spring Meeting in Paris (France), and became effective immediately.
The PSCI Principles for Responsible Supply Chain Management are foundational, setting members' expectations of suppliers, and form the base for all PSCI's tools and materials.
These Principles set the standard for governance and management systems, ethics, human rights, health and safety, and environmental business practices.
The Principles are adopted by all PSCI members and may be voluntarily supported by any business in the pharmaceutical and healthcare industry.
The countdown to the 2025 PSCI India Conference & Exhibition 2025 has begun, and the excitement is building!
The PSCI India team is thrilled to welcome all participants, speakers, presenters, and exhibitors for a dynamic event focused on building resilient supply chains through collaborative leadership.
The agenda is now live with sessions related to the PSCI Principles such as:
Download the agenda below
Join us for:
If you haven’t registered yet, now’s the time—secure your spot and be part of this impactful experience.
Please find the agenda for the PSCI Decarbonisation Summit 2025.
this agenda is subject to minor changes.
In the complex and highly regulated pharmaceutical and healthcare sectors, the ability to anticipate, withstand, respond to, and recover from disruptions is critical. This webinar will explore the fundamentals and strategic value of Business Continuity Management (BCM), with a particular focus on its alignment with supply chain risk management.
Attendees will gain insights into how BCM can be effectively integrated with incident and risk management systems to build resilience across operations. We’ll explore best practices for implementing a holistic, coordinated approach that ensures continuity in the face of global supply chain volatility, regulatory pressures, and emerging threats such as cyberattacks and natural disasters.
Why You Should Attend:
There will also be an opportunity to ask questions to our speaker during a dedicated Q&A session.
Speaker: Guy Stone
Head of Business Continuity Management for Roche Diagnostics since January 2025, responsible for defining and implementing the BCM Strategy for the Diagnostic Division of Roche and for providing central coordination as well as setting objectives and key results.
Associated recording available here
This one pager has been developed to provide a quick overview of the benefits of PSCI for suppliers to PSCI members.
We recommend that you share this with your suppliers whenever you are trying to provide a quick overview of PSCI & the benefits of engagement.
The PSCI Anti-trust Statement in English and Chinese.
2024 PSCI Chair Deirdre O’Reilly (Viatris) and 2024 Vice Chair Rob Williams (AstraZeneca) reflect on member-driven progress, shared achievements, and what’s next for the PSCI community. From deeper supplier engagement to an upgraded digital platform, hear how we’re continuing to build responsible pharmaceutical supply chains together.
An introduction deck for EQMS Ingenuity Private Limited
The PSCI presents its report on the use of Horseshoe Crab Blood for Endotoxin Testing in the Pharmaceutical Industry. The report provides insights into the context and complexities behind its use, pathways for better practices in the industry, an insight into the PSCI member companies’ use and next steps both the PSCI and others working in this ecosystem can take going forward.
The PSCI have been engaged on the topic of Horseshoe Crab Blood since 2023. Following the development of its position paper and convening through the working group. The PSCI has been discussing and exploring better pathways for better practices in the pharmaceutical industry and this report encouraging companies to minimize its use and seek alternatives for endotoxin testing in their supply chains.
Pharmaceutical companies use horseshoe crab blood to test medicines and vaccines for safety. The blood is a bright blue colour and has a special quality that makes it incredibly important to human health – it coagulates when exposed to bacterial endotoxins, which can cause sickness or even death in humans.
This project was funded through the projects workstream. Following report publication, the working group aims to publish further capability building resources to support good practices amongst PSCI members. While members are progressing on developing positions on the reduction or elimination of TAL and LAL in their supply chains, only few are be planning for the resilience of its supply.
The PSCI would like to extend a big thank you to Jay Bolden and Shah Shaid, members of the Horseshoe Crab Blood Working Group, the PSCI member companies that responded to the survey and all other contributors for the development of this year’s report.
The PSCI has launched our Supplier Partnership program to formally recognise and acknowledge suppliers’ commitment to responsible business practices. Becoming a PSCI Supplier Partner means committing to the PSCI Principles and contributing to a community committed to driving responsible value chains.
This presentation summarises the supplier partnership and provides an overview including:
The SAQ/Audit & Audit Guidance sub-team are pleased to publish the PSCI Pre-Audit Guidance Document Checklist. The checklist has been published as a separate document, previously found in the Audit Guidance as Annex 1.
The audit checklist summarizes important documents which the audit team may want to see in advance for audit preparation or want to review during the onsite audit visit. Depending on the type of supplier or the information provided as per SAQ, the list may be shortened (e.g. for service providers) or extended (e.g. for complex chemical or pharmaceutical manufacturers).
The checklist has been drafted using the current Full SAQ and Audit Report Template, suggesting documents that may be required for each principle (Governance & Management Systems, Ethics, Human Rights, Environment and Health & Safety) and corresponding question.
Climate change poses the greatest health threat of the 21st century and the pharmaceutical industry is a key contributor to global emissions due to complex product lifecycles and an energy-intensive value chain.
Many pharmaceutical companies have set ambitious, science-based decarbonization goals but only 20% of life sciences companies are on track to achieve net zero by 2050. Advancing decarbonization can be slow as companies wrestle with various pain points across the product lifecycle but delays in decarbonizing expose companies to risks across the stakeholder landscape. Decarbonizing the pharmaceutical industry is a core component of the PSCI’s Environment Principle and PSCI members realize the importance of working together and with their suppliers across the whole value chain.
There have been several whitepapers published on the topic of health sector decarbonization. This Playbook builds on that work to provide 24 detailed emissions reduction initiatives across 7 impact areas that map to the drug development lifecycle. The initiatives were assessed in terms of addressability, emission reduction potential, implementation timeline, upfront cost, and regulatory complexity, and intervention adoption timeframe.
We welcome your feedback. If you have any input on the Playbook, please contact us for consideration in future updates.
Revised January 2025
EXCEL VERSION WITH IMPORT SHEET (FOR ONLINE SAQ ONLY)
PSCI Self Assessment Questionnaire and Audit Report Template for Service Providers & General Manufacturers with import sheet.
For optimum functionality, we recommend using the most up to date version Microsoft Excel. If you only have access to Excel 2003 or earlier, we recommend using the Word version of the SAQ.
To access the offline abbreviated SAQ template and full version history of the SAQ, follow this link.
Content version 8.0, simplified the Categorisation of Suppliers (Category A, B or C), removed/ added a few questions, rewording some questions, December 2021. Import sheet incorporated July 2023.
Content version 9.0, full review and update of the SAQ. Section names and all questions revised to reflect the updated PSCI Principles, September 2024. Import sheet incorporated December 2024.
Full version template for Core Suppliers, External Manufacturers, Component and Material Suppliers are also available and come in multiple languages: Excel version full.
Please note: Content version 9.0 is only currently available in English with translations to follow soon.
The previous version of the SAQ is currently available in the following languages:
The following links provide SAQ Online instruction documents for:
THE PSCI PRESENTS ITS SECOND PUBLIC AUDIT FINDINGS REPORT, PROVIDING CONTINUED INSIGHTS INTO SUPPLIER RISKS, TRENDS, AND AREAS FOR IMPROVEMENT ACROSS THE PHARMACEUTICAL AND HEALTHCARE SUPPLY CHAIN.
This report shares our analysis of findings from PSCI audits available on our platform. Each year, we review these in line with the PSCI Principles to identify trends, highlight improvements, and share recommendations. Overall, this analysis provides deep insight into supplier practices within the pharmaceutical and healthcare industry and guides us on developing targeted resources and trainings.
The year’s report analyzes 1,776 findings from 180 supplier audits uploaded to the PSCI platform in 2023. These findings are categorized into 308 Management Systems findings, 7 Ethics findings, 169 Human Rights findings, 970 Health & Safety findings, and 322 Environment findings, offering a comprehensive overview of supplier practices and opportunities for improvement. The report includes a summary of findings across key topics, detailed breakdowns with recommended actions for suppliers, and links to relevant
What’s new this year?
The report reinforces the PSCI’s role as a catalyst for continuous improvement, equipping suppliers with tools and guidance to enhance practices and adapt to evolving demands. For more details, access the full report in the resource attached.
You're Invited: PSCI Global Supplier Conference 2025!
Get ready for a highly regarded line-up of expert speakers, insightful discussions, and actionable strategies to drive sustainability in pharmaceutical supply chains.
🔎 Featured Sessions:
📅 Download the full agenda below. To register, click here.
Don’t miss this opportunity to connect, learn, and contribute to the future of sustainable value chains. Secure your spot today and make every session count!
📩 Spread the Word: Forward this invitation to your colleagues.
If you have any questions, feel free to reach out at info@pscinitiative.org.
See you there!
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Jennifer Jiang | Partner | ERM
演讲嘉宾:
姜煜瑶 | 合伙人 | 伊尔姆
Topic: Scope 3 and Decarbonization
议题:范围三和脱碳
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Tracy Zhou | External Supply Chain EHS Manager | J&J
演讲嘉宾:
周穗菁 | 外部供应EHS经理 | 强生
Topic: The Management Maturity Model and Audit Findings
议题:PSCI治理与管理体系成熟度模型和审计发现项
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Lin Wang, Director, BSR China
演讲嘉宾:
王林,中国区总监,商业社会责任
Topic: China labor market practice and impact to business
议题:中国劳动市场的实践以及对商业的影响
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Jason Jiang| EHS Lead| GSK
Eric Peng| Regional Category Manager| Astra Zeneca
演讲嘉宾:
姜兴| EHS负责人 | 葛兰素史克
彭诗达| 亚太区采购经理 | 阿斯利康
Topic: Human Rights Audits Common Findings & CAPs
议题:劳动者权益审计常见发现项&整改方案| 劳动者权益话题分享
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Jing DONG | Managing Director | Peterson Solutions China
演讲嘉宾:
董菁 | 总经理 | 上海佩笙检测有限公司
Topic: Social Sustainability in Upstream Supply Chain via Responsible Sourcing Process
议题:通过负责任采购实现上游供应链中的社会可持续发展
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Gary Lu | APAC SHE & Sustainability Director | AstraZeneca
演讲嘉宾:
卢罡 | 亚太区安全健康环保及可持续发展总监 | 阿斯利康
Topic: China Green Power Project
议题:中国绿电项目
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 10, covering Management, Human rights & labor, and Decarbonization.
2024年中国供应商大会第一天于成都线下举行,覆盖管理体系、劳工、减碳议题。
Speaker:
Joyce Jiang| Sr Sustainability & Compliance manager, Global Procurement| Roche (Shanghai) Pharmaceuticals Consulting Co.,Ltd.
演讲嘉宾:
蒋南婧 | 全球采购-可持续发展与合规高级经理 | 罗氏(上海)医药咨询有限公司
Topic: Roche’s Journey to Decarbonise its Supply Chain - Challenges & Mitigation
议题:罗氏供应链的减碳之旅-挑战和行动
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The second session of the 2024 PSCI China Supplier Conference took place on September 11, covering Environment, Industrial Hygiene, General Safety, and Process Safety.
2024年中国供应商大会第二天于南京线下举行,覆盖环境、工业卫生、安全总览、过程安全议题。
Speaker:
Don Yu| BD Manager| BSI
演讲嘉宾:
于东 | 商务经理 | 英国标准协会
Topic: BSI Kitemark™ for minimized risk of antimicrobial resistance
议题:BSI与抗生素耐药性认证
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The second session of the 2024 PSCI China Supplier Conference took place on September 11, covering Environment, Industrial Hygiene, General Safety, and Process Safety.
2024年中国供应商大会第二天于南京线下举行,覆盖环境、工业卫生、安全总览、过程安全议题。
Speaker:
Xiaoyuan Zhang| EHS Technical Generalist | Jiuzhou Pharmaceutical Co.LTD
演讲嘉宾:
张孝远| EHS技术总工 | 浙江九洲药业股份有限公司
Topic: Jiuzhou Pharmaceutical Process Safety Management Case Sharing
议题:九洲药业过程安全管理案例分享
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The second session of the 2024 PSCI China Supplier Conference took place on September 11, covering Environment, Industrial Hygiene, General Safety, and Process Safety.
2024年中国供应商大会第二天于南京线下举行,覆盖环境、工业卫生、安全总览、过程安全议题。
Speaker:
William Zhu | Associate Director | WSP
演讲嘉宾:
朱人 | 副技术总监 | 科进
Topic: Statistical Analysis of Industrial Hygiene Monitoring Data
议题:工业卫生数据的统计分析
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The second session of the 2024 PSCI China Supplier Conference took place on September 11, covering Environment, Industrial Hygiene, General Safety, and Process Safety.
2024年中国供应商大会第二天于南京线下举行,覆盖环境、工业卫生、安全总览、过程安全议题。
Speaker:
Roderick Yuan| Technical Director | Shanghai STRM Technology Co., Ltd.
演讲嘉宾:
员文权| 技术总监 | 上海翊员科技有限公司
Topic: Mechanical integrity: Safety critical equipment that are of concern – and how we maintain those
议题:设备完好性-关键安全设施及检查、检测和预防性维护
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The first session of the 2024 PSCI China Supplier Conference took place on September 11, covering Environment, Industrial Hygiene, General Safety, and Process Safety.
2024年中国供应商大会第二天于南京线下举行,覆盖环境、工业卫生、安全总览、过程安全议题。
Speaker:
Carl Deng | Senior Process Safety Consultant | DEKRA Shanghai Office
演讲嘉宾:
邓胤 | 高级过程安全顾问 | DEKRA 德凯达管理咨询(上海)有限公司
Topic: Mechanical Equipment Ignition Risk Assessment (MEIRA)
议题:机械设备点火风险评估(MEIRA)
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮
The 2024 PSCI India Supplier Conference in Goa brought together over 700 participants for three days of insightful discussions and networking. Featuring keynote speeches from industry leaders, expert panels, PSCI’s first-ever exhibition, and an auditors' panel, the conference showcased the PSCI’s commitment to advancing responsible supply chain practices.
Thank you to everyone who joined us!
Please find the 2024 China supplier conference video recap for event highlights.
Thanks everyone for joining us!
This slide deck, presented by N T Prasad, Head EHSS Bangalore, Syngene International Limited during the "Process Safety" session at the PSCI India Supplier Conference 2024 in Goa, India.
This resource covers key trends, challenges, and best practices relevant to PSCI members and suppliers, the resource is designed to enhance understanding, with actionable strategies and case studies relevant to India and the broader region. Perfect for those looking to deepen their knowledge in sustainable supply chain management and industry collaboration.
During 2024 we will be updating/replacing The Link – the online platform by which we communicate with members and share audits.
We have today launched an open tender exercise to find the best provider. The attached documents have been sent to a small group of selected bidders.
We would welcome the widest possible range of submissions.
Supporting documents:
PSCI Link 2.0 RFP - via the download button below.
Link system map
Link 2.0 user requirements
For ease, please note the following:
Address for correspondence is: LinkRFP@pscinitiative.org
Please confirm your interest to participate by: 10th May 2024
Bid submission deadline: 31st May 2024
PSCI has conducted a materiality review for the sector every few years (2013, 2017, 2020). We are pleased to publish our latest assessment, this time applying a double materiality framework. The assessment is specifically focused on supply chains for the Pharmaceutical & Healthcare industries, and takes into account member views and a range of outside sources. (For more detail about the double materiality approach, please see here.)
The initial materiality was conducted in Summer 2023 to support development of the new strategy. This has since been updated to take into take into account the final CSRD and to add an additional checking stage to the scoring and issues list.
We encourage members, suppliers and our other stakeholders and invited to read the report and use it as an input to their own planning processes.
For anyone with comments to the report, please contact Rosie.Towe@Carnstone.com.
As part of the PSCI's goal to promote consistency, quality, and innovation in all aspects of supplier evaluation and audit, we analyze PSCI audits conducted and uploaded on our shared platform on an annual basis. This report shows what we’ve found by analyzing 3,500 findings from 339 PSCI audits conducted during 2020-2022 and uploaded to the PSCI platform by the end of 2022.
We are specifically interested in understanding how the findings are distributed across the five areas of the PSCI Principles, our blueprint for responsible supply chain management & foundation of our tools. This analysis gives us visibility over how findings are changing over time and our impact on the industry.
This report, the first we have shared publicly, shows the results of a deep dive analysis into each area, focusing on 1,955 findings from 195 PSCI audits uploaded by the end of 2022. These findings reveal key risks, areas for improvement, and opportunities for PSCI members to support their suppliers to improve their practices.
This analysis also provides a high-level understanding of regional differences and levels of maturity, allowing us to provide regionally specific support on hot topics through our capability building work and begin to understand our own impact on the industry.
Speaking of the report's publication, Dorota Wiacek-Trojanowska, Audit Committee member says,
The annual analysis of audit results carried out by the PSCI not only provides information enabling the identification of the most important challenges faced by both PSCI members and suppliers, but also shows new challenges that we must be able to face. The audit results give us the opportunity to collect and analyze information on the biggest challenges in ensuring a sustainable supply chain in the whole pharma sector.
Please find the India Supplier Conference 2023 Video Recap for highlights of the event. Thanks to everyone who joined us for this exciting event!
Bylaws of the Pharmaceutical Supply Chain Initiative. Last updated in January 2024.
The updated PSCI Principles were adopted on 23 May 2023.
To help members and suppliers reflect the changes in their policies, codes, processes, and practices, please find below a short deck outlining the main changes to the Principles. This includes a one-pager at the front summarizing the key updates.
As a reminder, you may download the full document comparing the old and new versions side-by-side here.
The refreshed Principles are in effect. The first check-in will take place during the annual member calls in March 2024. All PSCI member companies are expected to have integrated the revised PSCI Principles by November 2024.
The PSCI has published our 2022 Annual Report, summarizing a year of growing connections and clear movement towards achieving our shared vision of excellence in safety, environmental, and social outcomes across the global pharmaceutical and healthcare supply chain.
Key developments in 2022 include
Vicki Stone-Bjarup, 2022 Chair of the PSCI, says,
Sectors around the world are working to tackle the sustainability challenges of their industry. This report transparently shares the successes that PSCI members have achieved for our sector over the last year, as well as the journey left to take. There is still a long way to go, but we are so proud to be a part of the progress being made in the pharmaceutical industry. Our focus remains to support our members to build responsible supply chains.
Read about these and more in the report below.
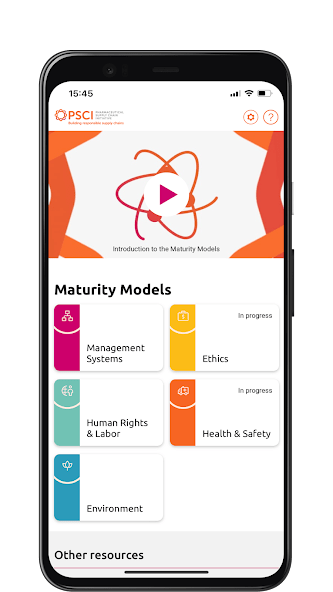
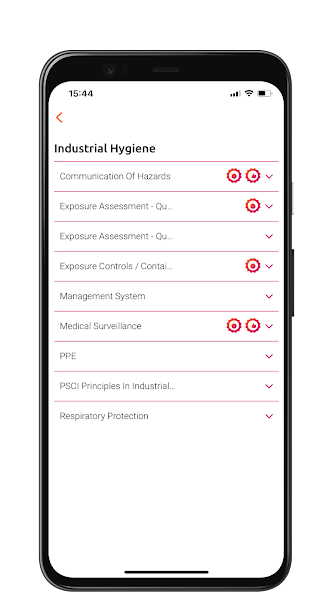
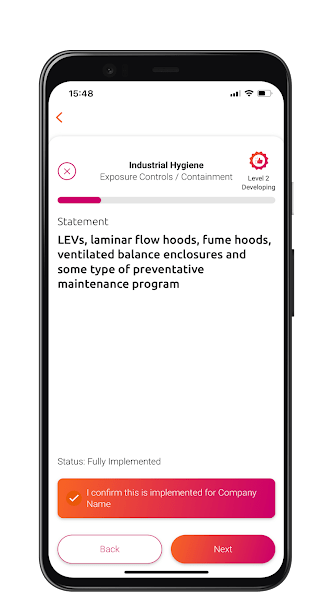
The PSCI Maturity App provides an interactive experience of the PSCI Maturity Models. Available on Android and iOS.