Musi River WWW 2023: Presentation by PiE/AMR
Presentation given by Balaji G. at the Musi River Wastewater Workshop 2023.
Access our full library of resources, register for events and webinars and share audits and other assessments
Our resource library is made available freely to all in order to advance responsible supply chain practices. Here you will find information and guidance on responsible supply chain issues and the way that the PSCI works. If you have any suggestions for improvements or additions to our resources please contact us.
Presentation given by Balaji G. at the Musi River Wastewater Workshop 2023.
Decarbonizing the pharmaceutical industry in line with global efforts is a core component of PSCI’s environmental principle. Action is needed and PSCI members realize the importance of working together with their suppliers, often the majority of the footprint across the whole value chain.
This is why we're pleased to launch the PSCI Decarbonization Pathway, a tool that the industry can use to move towards a net zero value chain, in line with global goals such as the COP Paris Agreement and UN Sustainable Development Goals. The maturity model provides a clear pathway for suppliers to develop their capability and response towards reducing their carbon emissions.
PSCI Members, through their Decarbonization team and partnerships with leading organisations, will continue to provide the resources that suppliers will need in this journey.
Version: v3 August 2025
In this video, the PSCI reiterates its commitment to supporting our members to understand human rights practices in pharmaceutical supply chains and outlines the resources available to help make it happen across the industry.
Together with our members, the PSCI understands that society and business are best served by responsible business behaviors and practices. With our training materials, conferences, and tools, we aim not only to ensure the upholding of our Principles, but also to support all our members in implementing the United Nations’ Guiding Principles on Business and Human Rights in their supply chain and improving their human rights impacts around the world.
The first session of the 2022 PSCI China supplier conferences took place virtually on 6 September and covered Ethics, Labor, Management systems.
2022年中国供应商线上会议第一场于9月6日在线上举行,覆盖伦理,劳工,管理议题。
Speaker: Roderick | Technical director | Shanghai STRM Technology Co., Ltd.
演讲嘉宾:员文权 | 技术总监 | 上海翊员科技有限公司
Topic: Business continuity plan
议题:业务连续性计划
Watch the recording here
点击此处查看回放
To access the slides, please click "DOWNLOAD". 下载会议PPT,请点击下方DOWNLOAD按钮·
The third session of the 2022 PSCI India supplier conferences took place in-person on the 23rd of September and covered Human Rights & Labor and Ethics.
The full slide deck is available for download below.
The presentations covered are as follows:
The second session of the 2022 PSCI India supplier conferences took place in-person on the 22nd of September and covered Audit, Environment, Scope 3, PiE/AMR
The full slide deck is available for download below.
The presentations covered are as follows:
The first session of the 2022 PSCI India supplier conferences took place in-person on the 21st of September and covered Safety and Process Safety Management.
The full slide deck is available for download below at "View More".
The presentations covered are as follows:
The third session of the 2022 PSCI China supplier conferences took place virtually on 8 September and covered PiE / AMR and Environment.
Please click “DOWNLOAD” to access to session 3's deck.
The third session includes:
Management of change
Genping Zou | APAC region HSE manager | Bayer
EPSC : process safety hazards for pharma operation units
Li Liu | EHS manger | BI China EHS&S
Safety maturity model
Barry Bai | Sr. HSE manager | External Manufacturing China Elanco
Road transportation of dangerous goods regulations (JT-617)
Tina Wang | Project director | Dow & Ann Consulting (Shanghai) Ltd.,
How to complete a high-quality equipment containment performance test
Vincent Zhang | IH service leader | Joinhand Consulting Co., Ltd.
IH modelling and its application in quantitative exposure assessmen
Wenjia Xu | Associate Director, Global Safety & Environment | MSD
——————————————————————————————————————
变更管理
邹根平 | 亚太地区HSE经理 | 拜耳
EPSC:制药单元操作中的工艺安全危害及其典型的控制措施
刘立 | EHS 经理 | 勃林格殷格翰
安全成熟度模型
白大明 | HSE高级经理 | 礼来
危险品道路运输规定(JT-617)- 化学品车辆合规性检查
王文 | 项目总监 | 道安咨询(上海)有限公司
如何完成一次高质量的密闭测试
张佳维 | 工业卫生咨询业务负责人 | 章含管理咨询(上海)有限公司
职业卫生模拟及其在定量接触评估中的应用
徐文嘉|副总监,全球安全与环境|默沙东
The first session of the 2022 PSCI China supplier conferences took place virtually on 6 September and covered PiE / AMR and Environment.
Please click “DOWNLOAD” to access to session 1's deck.
The first session includes:
Business continuity plan
Roderick | Technical director | Shanghai STRM Technology Co., Ltd.
PSCI Audit program – practical guidance
Catherine Zhang | Head of HSE experts APAC | PSCI China sub team lead | Bayer
Supplier sharing: CAPR formulation & common audit findings
Ying Lin | EHS & Assistant GM | Zhejiang Raybow Pharmaceutical Co. LTD
Danhua Xu | EHS associate manager | Tianyu Pharmaceutical Co. LTD
The new Data Privacy Law in China
Chris Zhang | Senior partner | Dentons Law Firm
New labor codes
Minnie Mai | Senior technical manager | TUV
——————————————————————————————————————
业务连续性计划
员文权 | 技术总监 | 上海翊员科技有限公司
PSCI审计项目分享
张晓花 |亚太区健康安全环境专家组负责人 | PSCI 中国小组负责人 | 拜耳
供应商实践分享: 不同审计体系共性发现项CAPR制定
林迎 | 总经理助理(分管EHS)| 浙江瑞博制药有限公司
许丹华 | EHS副总经理 | 临海天宇药业有限公司
中国新数据隐私法
张勇 | 高级合伙人 | 大成律师事务所
新劳工法规
麦璐 | 高级技术经理 | 德国莱茵
The second session of the 2022 PSCI China supplier conferences took place virtually on 7 September and covered PiE / AMR and Environment.
Please click “DOWNLOAD” to access to session 2's deck.
The second session includes:
Mass balance work tool
Annie Feng | Principal Consultant | Arcadis
Supplier practice sharing: toxicant minimization in API wastewater
Qinfan Zhang | Director China | Enviolet GmbH
Candy Chen | EHS manager | Zhejiang Hisoar Chuannan Pharma Co., Ltd.
Waste minimization - application of flow chemistry
Jian Tao | Vice general manager | Center of Flow & Continuous Technology (CFCT) | Asymchem
Environmental protection project management
Qingpeng Zhu | Environment protection supervisor | Pharmaron
ESG and sustainability reporting
Ellen Zhang | Senior manager of expert support services, head of China Office | Enhesa
GHG emissions accounting (Supplier Scope 1 & 2)
Zoey Tang | EHS Consultant | Golder
——————————————————————————————————————
2022PSCI线上供应商会议(中国)第二场在9月7日举行,会议内容包括环境中的药物 / 抗生素抗药性,环境。
会议PPT请在下方点击“DOWNLOAD”下载。
第二场会议具体包括:
确定药物生产过程API损失的预测环境浓度
冯婉华博士| 首席顾问 | 凯谛思
供应商实践分享: API废水排放-废水中的毒物减少
张庆凡 | 中国大区总经理 | 恩维勒
陈燕芳 | EHS经理 | 浙江海翔川南药业有限公司
废弃物减少-废弃物减少- 流动化学的应用
陶建 | 副总经理 | 凯莱因医药集团-连续科学技术中心
环保工程建设管理
朱庆鹏 | EHS主管 | 康龙化成(天津)药物制备技术有限公司
ESG与可持续发展报告分享
张赟 | 专家支持服务高级经理,中国办公室负责人 | 音和环保咨询(上海)有限公司
温室气体核算
汤淋麟 | EHS咨询顾问 | WSP Golder
The PSCI is pleased to introduce the Maturity Models in this informative video. The Maturity Models are a very useful tool for suppliers to evaluate their current practices and plot a course for improvement. They allow users to evaluate their company practices against the PSCI Principles and signpost the user to the resources needed to progress.
The PSCI is pleased to introduce the Maturity Models in this informative video. The Maturity Models are a very useful tool for suppliers to evaluate their current practices and plot a course for improvement. They allow users to evaluate their company practices against the PSCI Principles and signpost the user to the resources needed to progress.
This version is subtitled
Today the PSCI published its 2021 Annual Report, summarizing a year of exceptional advancement towards achieving our shared vision of excellence in safety, environmental, and social outcomes across the global pharmaceutical and healthcare supply chain.
Key developments in 2021 include:
Manjit Singh, 2021 PSCI Chair and Associate Director of Corporate Sustainability at Centrient Pharmaceuticals says,
2021 was a year of spectacular growth for the PSCI in membership and activity. Despite the second year of the Covid-19 pandemic, every aspect of the PSCI's program developed and grew from our shared audits to our supplier capability interactions. I have appreciated the professional and energetic contribution of my fellow PSCI members and watched with great pride the growth of the organization and the impact it has upon our entire industry.
You can read about these advancements and more in the report below.
We welcome comments and encourage you to share this report with your networks using the hashtag #PSCIAnnualReport.
The Musi River flows through Hyderabad in Telangana State, Southern India. It is fed by the world-heritage Hussein Sagar lake, also called the “heart of the world”, and used to be enjoyed by locals and tourists alike for its beauty and amenity. Yet, there have been numerous reports of polluted effluents and poor wastewater management practices affecting the lake and the Musi River watershed, impacting local communities over many years.
For World Water Day 2021 (22 March), we published a statement outlining our support for the State government's initiative and our plans to help. We aimed to use our influence to engage with suppliers of PSCI members in Telangana to promote better wastewater practices and encourage positive change. Our aim is that every single supplier with a connection to a PSCI member should be visited, audited or contacted directly to promote the best technologies for wastewater management.
One year later, we're pleased to share an update on progress made. Since March 2021, we have
Our aim is that all suppliers to PSCI members should provide information about their wastewater treatment practices, either via member visit, questionnaire, or a full site audit shared on our platform.
To continue this work, we will
We hope to contribute to restoring the Musi River to its former health.
This document addresses the common questions suppliers have around the PSCI audit program. The key points include:
The Audit Committee has recently conducted a light refresh on the SAQ/Audit Excel template, and the Audit Guidance document. The new versions are now available on the Link.
This webinar covered the following topics:
We were delighted to be joined by:
The recording is available here.
The slides used during the webinar are available for download below.
Our final newsletter of 2021 reflects on our virtual AGM, successful Supplier Conferences, and the launch of our position statement on Human Rights.
At the PSCI, we’re committed to supporting all our members to implement the United Nations’ Guiding Principles on Business and Human Rights (UNGPs) as part of our mission to drive excellence in safety, environmental, and social outcomes across the pharmaceutical and healthcare supply chain.
That’s why for Human Rights Day (10 December), we’re excited to launch our new Position Statement, which provides an overview of the role we play in helping our members and suppliers align their practices with the UNGPs through the PSCI Principles for Responsible Supply Chain Management and our resources.
With our training materials, webinars, and tools we aim not only to ensure the upholding of our Principles, but also to support all our members in implementing the UNGPs across their entire supply chains.
You can read the full statement below.
EXCEL VERSION
PSCI Self Assessment Questionnaire and Audit Report Template for Core Suppliers, External Manufacturers, Component and Material Suppliers
For optimum functionality, we recommend using the most up to date version Microsoft Excel. If you only have access to Excel 2003 or earlier, we recommend using the Word version of the SAQ.
Content Version 6, February 2019
Content Version 7, remote audit options added, October 2020
Content version 7.1, small technical fixes, including correctly referencing finding type on CAP tab, showing suppliers responses to topic areas that are not audited, May 2021
Content version 7.2, fix formating issue of finding classifications on the Company Specific Questions tab, June 2021
Content version 8.0, simplified the Categorisation of Suppliers (Category A, B or C), removed/ added a few questions, rewording some questions, December 2021
Content version 9.0, full review and update of the SAQ. Section names and all questions revised to reflect the updated PSCI Principles, September 2024.
Abbreviated version template for Service Providers & General Manufacturers are also available: Excel version.
The SAQ is currently available in the following languages:
EXCEL VERSION
Abbreviated PSCI Self Assessment Questionnaire & Audit Report Template for Service Providers & General Manufacturers
For optimum functionality, we recommend using the most up to date version Microsoft Excel. If you only have access to Excel 2003 or earlier, we recommend using the Word version of the SAQ.
Content Version 6, February 2019
Content Version 7, remote audit options added, October 2020
Content version 7.1, small technical fixes, May 2021
Content version 7.2, fix formating issue of finding classifications on the Company Specific Questions tab, June 2021
Content version 8.0, simplified the Categorisation of Suppliers (Category A, B or C), removed/ added a few questions, rewording some questions, December 2021
Content version 9.0, full review and update of the SAQ. Section names and all questions revised to reflect the updated PSCI Principles, December 2024.
Full version template for Core Suppliers, External Manufacturers, Component and Material Suppliers are also available and come in multiple languages: Excel version.
The second session of the 2021 PSCI China supplier conferences took place virtually on 10 September and covered PiE / AMR.
The full recording of the session is available here. Slides are available for download below.
2021PSCI线上供应商会议(中国)第二场在9月10日举行,会议内容包括环境中的药物 / 抗生素抗药性。
本次会议的完整视频请点击链接 (提取码 6666) 获取。会议PPT请在下方点击“DOWNLOAD”下载。
The second session includes: 第二场会议具体包括:
This is an explainer video on the PSCI Topic Teams with English subtitles.
This is a short video explaining the Topic Teams of the PSCI.
The fourth session of the 2021 PSCI China supplier conferences took place virtually on 17 September and covered Safety, Process Safety Management (PSM) .
The full recording of the session is available here. Slides are available for download below.
2021PSCI线上供应商会议(中国)第四场在9月17日举行,会议内容包括安全 & 过程安全管理 。
本次会议的完整视频请点击链接 (提取码 txg2 ) 获取。会议PPT请在下方点击“DOWNLOAD”下载。
The fourth session includes: 第四场会议具体包括:
LOTO best practice
Frank Deng, EHS Manager, TÜV Rheinland
上锁挂牌的最佳实践 (50mins 分钟)
邓柱明,EHS经理,德国莱茵
Local government regulations and best practices around hot work and confined space
Shiwei Kong, EHS Head, Zhejiang Langhua Pharmaceutical
动火作业和受限空间作业:各级政府要求与企业最佳实践 (50mins 分钟)
孔识卫, EHS总监, 浙江朗华制药有限公司
Preventive Maintenance for safety equipment
Shuquan Chen, Supplier Operations EHS Manager, Pfizer
安全设备预防性维护 (50mins 分钟)
陈树权,供应商运营EHS经理,辉瑞
The third session of the 2021 PSCI China supplier conferences took place virtually on 16 September and covered Management systems, Industrial hygiene.
The full recording of the session is available here. Slides are available for download below.
2021PSCI线上供应商会议(中国)第三场在9月16日举行,会议内容包括环境中的管理体系 & 工业卫生 。
本次会议的完整视频请点击链接 (提取码 jg38) 获取。会议PPT请在下方点击“DOWNLOAD”下载。
The third session includes: 第三场会议具体包括:
The Practice of Dual-system in Pharmaceutical Enterprise
Guoqiang Peng, EHS Manager Assistant, Hangzhou Zhongmei Huadong Pharmaceutical
双重预防体系(双体系)在制药企业中的运用
彭国强,EHS经理助理,杭州中美华东医药股份有限公司
Pharma wastewater API testing
Congyi Xu, Senior regulatory consultant, CIRS
中国《新化学物质环境管理办法》解析
许丛艺, 资深化学品法规咨询师, 瑞旭集团
Occupational exposure band and control technology in pharmaceutical industry
Chengyin Guo, Deputy-director of EHS, Raybow Pharma
职业卫生分级管控及密闭技术在制药企业中的应用
郭成寅,EHS副总监,瑞博制药
The first session of the 2021 PSCI China supplier conferences took place virtually on 9 September and covered Introduction, Human Rights & Labor, Environment.
The full recording of the session is available here. Slides are available for download below.
2021PSCI线上供应商会议(中国)第一场在9月9日举行,会议内容包括PSCI 最新发展、劳工及环境。
本次会议的完整视频请点击链接 (提取码 9m1p) 获取。会议PPT请在下方点击“DOWNLOAD”下载。
The first session includes: 第一场会议具体包括:
Recent developments within PSCI
Manjit Singh, PSCI Chair, Associate Director - Corporate Sustainability, Centrient
PSCI最新动态
Manjit Singh, PSCI主席,可持续发展副总监,灿盛制药
Audit Committee update: remote audit, audit sharing and supplier self-initiated audit
Kelley Jiang, Head HSE TPRM Operational Excellence Global HSE, Novartis
审核委员会最新内容: 远程审计、共享审计报告与供应商自发PSCI 审核
江戎, 大中国区供应商健康安全环境风险管理主管, 诺华制药
Common findings in PSCI audits under Labor section
Minnie Mai, Senior Technical Manager,TÜV Rheinland
审核劳工部分常见发现项
麦璐, 高级技术经理,德国莱茵
3060 carbon targets and latest government regulations
Stone Huang, lawyer and certified safety engineer, Jin Mao Law Firm 3060双碳目标制定背景双碳目标制定背景及及最新相关法规政策探讨
黄启荣,律师/注册安全工程师, 金茂律师事务所
The issue of Pharmaceuticals in the Environment (PiE) is an increasing public concern. While manufacturing losses represent a small percentage of the overall API mass entering the environment, the PSCI is committed to addressing localized risks that may occur from pharmaceutical manufacturing operations. Many companies are requiring that their facilities – and their major suppliers – establish estimates for API mass loss and the resulting Predicted Environmental Concentration (PEC) in the water body that ultimately receives a facility’s wastewater discharge.
The PSCI-endorsed method for estimating API mass loss is to follow a mass balance approach where the API inputs, outputs, and losses are accounted for, allowing the mass entering wastewater to be estimated. The PSCI has developed a set of detailed guidance documents on how to approach the mass balance calculations, including this presentation which provides an overview of:
We were delighted to be joined on this webinar by:
The recording is available here.
The slides used during the webinar are available for download below.
Participants are invited to share any feedback about this webinar by completing this survey.
The following document provides guidance on potential techniques, methodologies, and available data sources for the calculation of dilution factors. The correct approach to follow is dependent on specific aspects of the risk assessment to be conducted. The PSCI does not advocate a single correct approach or data source, but aims to provide information to help risk assessors design assessments suitable for their requirements.
The contents of this document has been kindly shared by the IAI PiE Task Force.
The following document provides guidance on potential techniques, methodologies, and available data sources for sampling and analysis of pharmaceutical industry wastewater. The correct approach to follow is dependent on specific aspects of the risk assessment to be conducted. The PSCI does not advocate a single correct approach or data source, but aims to provide information to help risk assessors design assessments suitable for their requirements.
The contents of this document has been kindly shared by the IAI PiE Task Force.
Please download to access the agenda for the three-day virtual event.
A clear process has been defined to ensure PSCI Audits are consistent and rigorous. A suite of tools are available to support and guide through the process. Follow the link to read more about:
In order to ensure quality and integrity, PSCI Audits are carried out either by qualified internal auditors working at PSCI member companies or by professional and independent third party audit firms.
PSCI has approved fifteen professional, independent third party audit firms to perform PSCI Audits.
This is an explainer video on the PSCI Governance Structure with English subtitles.
This is a short video explaining the Governance Structure of the PSCI.
Anti-bribery and corruption is one of the issues under the PSCI Principles. The PSCI has commissioned an external benchmark on anti-bribery and corruption (ABC) standards and trends in supply chain. This report has been created to help our member companies and suppliers strengthen the ABC risk management practice in their supply chain.
Background: Regulations around anti‐corruption have been tightened and stakeholder expectations have increased. PSCI wants to encourage and support members and suppliers to continually enhance their anti‐corruption program for compliance with the latest guidelines issued by different regulatory authorities around the globe. Organizations are expected to develop proactive, risk‐based compliance programs that are tailored to the specific risk profile and are reasonably designed to prevent them and their employees/agents/suppliers from engaging in bribery or corrupt acts.
A short explainer video on the PSCI Principles for Responsible Supply Chain Management with English subtitles.
A short explainer video on the PSCI Principles for Responsible Supply Chain Management.
We are very pleased to announce that the 2020 PSCI Annual Report was launched today, summarizing the progress and achievements we have made in advancing responsible supply chain practices within the pharmaceutical and healthcare industries over the past year.
Key developments in 2020 include:
2020 was an extraordinary year, full of large-scale professional challenges for our industry and its supply chain, not to mention the personal challenges we all faced in our own lives. We are proud of the way the PSCI adjusted to these challenges, showing remarkable agility. It is clear that the importance of responsible supply chain practices cannot be overstated and we are pleased to be able to drive positive change across the industry through our collaborative efforts.
We welcome comments and encourage you to share this report with your networks.
Since 2016, a group of PSCI members have been working together to standardize their environmental data request to suppliers through a common set of questions, known as the PSCI Environmental Survey. The benefit is that suppliers should be receiving a common set of questions, but currently companies collect the data using different routes: some use an Excel file, and some a third-party platform.
The PSCI has now built the Environmental Survey into the PSCI platform for suppliers (called The Link). Each of the PSCI’s 50 members will now be able to use that platform to collect data from suppliers and we anticipate that many will do so. We hope that this will streamline and simplify your reporting of environmental and carbon emissions to your customers as one reply can now be shared with many customers.
Watch this webinar to learn more about the new platform and how to use it. You'll hear about:
We were delighted to be joined by Alejandro Fiocco, Partner at Carnstone and Secretariat for the PSCI Environment Team.
The recording is available here. (Please refresh the page if you cannot play the video.)
The slides used during the webinar are available for download below.
Participants (including registrants who were unable to attend) are invited to share feedback about this webinar here.
Please also note that guidance about the Environmental survey is available here (the excel version is accessible here).
More great publicity for the PSCI with thanks to Dan Caldwell (Johnson & Johnson) and Steve Brooks (AMR Alliance) who recently recorded this CPhI podcast on pharmaceuticals in the environment (PiE) and anti-microbial resistance (AMR) and what the industry is doing to help address the challenge.
The PSCI has published a PEC:PNEC calculator tool for manufacturers to use to calculate how to meet safe levels of discharge for active pharmaceutical ingredients (APIs) from their sites.
Image credit: CPhI
This document is a step by step guidance on using the PSCI Environmental survey for suppliers on the PSCI platform. If you have any further questions, please do not hesitate to contact us.
This file contains the latest version of the PSCI Environmental survey for suppliers in Excel. The scoring for each question is detailed and questions are mapped across to the previous version.
Our April newsletter reflects recent activities including our support to the Musi River Revitalization Initiative, the publication of our Benchmarking Survey Insights Report, and our outreach campaign on the vital issue of anti-microbial resistance. It also highlights our upcoming plans and events, such as our Virtual Spring Meeting, and external developments relevant to pharmaceutical supply chains.
At the PSCI, we’re committed to doing everything in our power to limit that the spread and growth of two emerging threats – Pharmaceuticals in the Environment (PiE) and Anti-microbial Resistance (AMR). That’s why for World Health Day, we’re excited today to launch our new Position Statement, which provides a clear explanation of the critical role we play in helping our supplier partners improve their manufacturing practices. The statement has been written to highlight the relevance of the PiE issue along the supply chain, to articulate the PSCI's position on this high profile topic, and to help answer stakeholder inquiries. It includes an overview of all PSCI resources on PiE and AMR-related issues.
Updated “draft” PSCI surface water PEC:PNEC calculator tool
Version: 23 March
Supplier Self-initiated Audit Programme (SSIA) is a pilot programme led by PSCI Audit Committee to encourage suppliers to take a proactive, risk-based approach and self-initiate PSCI audits for their facilities.
With the pilot programme this year, the PSCI will pay for a new Supplier Self-initiated Audit of the supplier facility. The audit should be completed by a PSCI approved audit firm and shared with PSCI members on the Link. Any supplier to PSCI Member can apply to participate in SSIA.
This document for suppliers explains:
Members are welcome to share the brochure with suppliers you would like to see conducting a PSCI audit.
If you are interested in learning more about the programme, please contact Audit Committee Secretariat Blake Zheng for more information. Thank you.
In 2020, we invited the PSCI members to participate in our third formal Member Benchmarking Survey and we are pleased to present the results in this Insights Report. We have used the data collected over the years to share year-on-year trends, as well as provide a snapshot from 2020.
This report paints a picture of rising engagement and increasing focus on responsible procurement. We hope that you find it useful and insightful.
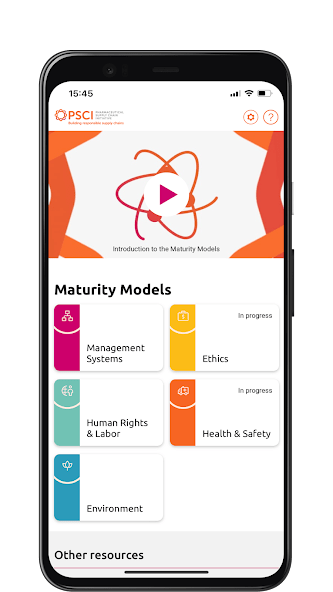
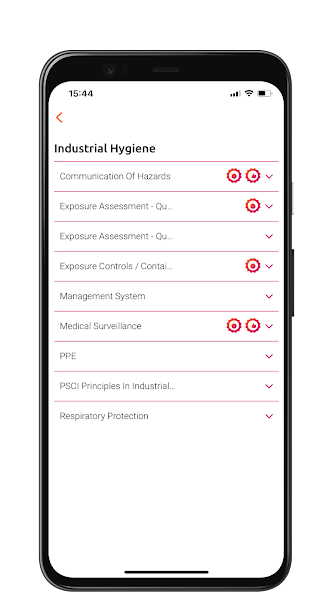
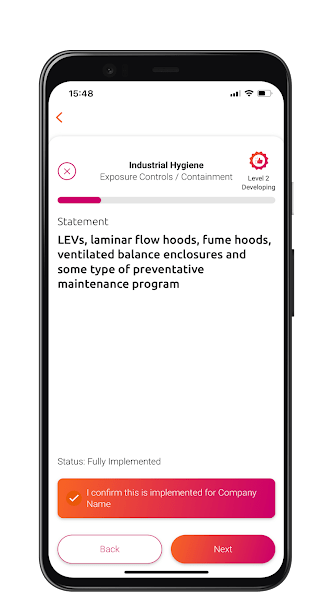
The PSCI Maturity App provides an interactive experience of the PSCI Maturity Models. Available on Android and iOS.